Bevacizumab for newly diagnosed pleural mesothelioma in the Mesothelioma Avastin Cisplatin Pemetrexed Study (MAPS): a randomised, controlled, open- label, phase 3 trial - The Lancet

Bevacizumab plus capecitabine versus capecitabine alone in elderly patients with previously untreated metastatic colorectal cancer (AVEX): an open-label, randomised phase 3 trial - The Lancet Oncology
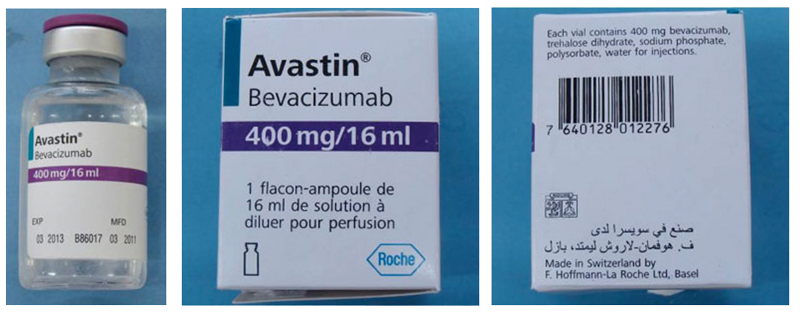
Genentech: Statements | Genentech Statement on Counterfeit Drug Labeled as Avastin® (Bevacizumab) in the United States